
Ideal-dilute solutions
 المؤلف:
Peter Atkins، Julio de Paula
المؤلف:
Peter Atkins، Julio de Paula
 المصدر:
ATKINS PHYSICAL CHEMISTRY
المصدر:
ATKINS PHYSICAL CHEMISTRY
 الجزء والصفحة:
ص145-148
الجزء والصفحة:
ص145-148
 2025-11-13
2025-11-13
 69
69
Ideal-dilute solutions
In ideal solutions the solute, as well as the solvent, obeys Raoult’s law. However, the English chemist William Henry found experimentally that, for real solutions at low concentrations, although the vapour pressure of the solute is proportional to its mole fraction, the constant of proportionality is not the vapour pressure of the pure sub stance (Fig. 5.15). Henry’s law is:
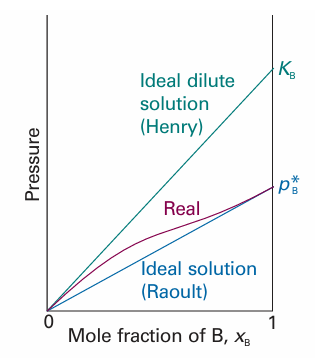
Fig. 5.15 When a component (the solvent) is nearly pure, it has a vapour pressure that is proportional to mole fraction with a slope pB * (Raoult’s law). When it is the minor component (the solute) its vapour pressure is still proportional to the mole fraction, but the constant of proportionality is now KB (Henry’s law).
pB=xBKB
In this expression xB is the mole fraction of the solute and KB is an empirical constant (with the dimensions of pressure) chosen so that the plot of the vapour pressure of B against its mole fraction is tangent to the experimental curve at xB=0. Mixtures for which the solute obeys Henry’s law and the solvent obeys Raoult’s law are called ideal-dilute solutions. We shall also label equations with a superscript ° when they have been derived from Henry’s law. The difference in behaviour of the solute and solvent at low concentrations (as expressed by Henry’s and Raoult’s laws, respectively) arises from the fact that in a dilute solution the solvent molecules are in an environment very much like the one they have in the pure liquid (Fig. 5.16). In contrast, the solute molecules are surrounded by solvent molecules, which is entirely different from their environment when pure. Thus, the solvent behaves like a slightly modified pure liquid, but the solute behaves entirely differently from its pure state unless the solvent and solute molecules happen to be very similar. In the latter case, the solute also obeys Raoult’s law.
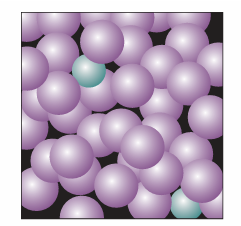
Fig. 5.16 In a dilute solution, the solvent molecules (the purple spheres) are in an environment that differs only slightly from that of the pure solvent. The solute particles, however, are in an environment totally unlike that of the pure solute.
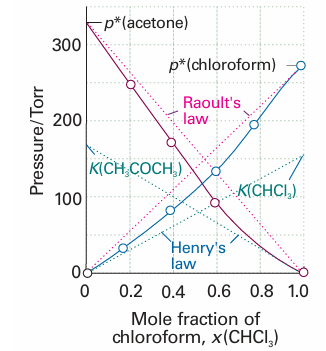
Fig. 5.17The experimental partial vapour pressures of a mixture of chloroform (trichloromethane) and acetone (propanone) based on the data in Example 5.3. The values of Kare obtained by extrapolating the dilute solution vapour pressures as explained in the Example.

For practical applications, Henry’s law is expressed in terms of the molality, b, of the solute, pB =bBKB Some Henry’s law data for this convention are listed in Table 5.1. As well as providing a link between the mole fraction of solute and its partial pressure, the data in the table may also be used to calculate gas solubilities. A knowledge of Henry’s law constants for gases in blood and fats is important for the discussion of respiration, especially when the partial pressure of oxygen is abnormal, as in diving and mountaineering, and for the discussion of the action of gaseous anaesthetics.

We inhale about 500 cm3 of air with each breath we take. The influx of air is a result of changes in volume of the lungs as the diaphragm is depressed and the chest expands, which results in a decrease in pressure of about 100 Pa relative to atmospheric pressure. Expiration occurs as the diaphragm rises and the chest contracts, and gives rise to a differential pressure of about 100 Pa above atmospheric pressure. The total volume of air in the lungs is about 6 dm3, and the additional volume of air that can be exhaled forcefully after normal expiration is about 1.5 dm3. Some air remains in the lungs at all times to prevent the collapse of the alveoli. A knowledge of Henry’s law constants for gases in fats and lipids is important for the discussion of respiration. The effect of gas exchange between blood and air inside the alveoli of the lungs means that the composition of the air in the lungs changes throughout the breathing cycle. Alveolar gas is in fact a mixture of newly inhaled air and air about to be exhaled. The concentration of oxygen present in arterial blood is equivalent to a partial pressure of about 40 Torr (5.3 kPa), whereas the partial pressure of freshly inhaled air is about 104 Torr (13.9 kPa). Arterial blood remains in the capillary passing through the wall of an alveolus for about 0.75 s, but such is the steepness of the pressure gradient that it becomes fully saturated with oxygen in about 0.25 s. If the lungs collect fluids (as in pneumonia), then the respiratory membrane thickens, diffusion is greatly slowed, and body tissues begin to suffer from oxygen starvation. Carbon dioxide moves in the opposite direction across the respiratory tissue, but the partial pressure gradient is much less, corresponding to about 5 Torr (0.7 kPa) in blood and 40 Torr (5.3 kPa) in air at equilibrium. However, because carbon dioxide is much more soluble in the alveolar fluid than oxygen is, equal amounts of oxygen and carbon dioxide are exchanged in each breath. A hyperbaric oxygen chamber, in which oxygen is at an elevated partial pressure, is used to treat certain types of disease. Carbon monoxide poisoning can be treated in this way as can the consequences of shock. Diseases that are caused by anaerobic bacteria, such as gas gangrene and tetanus, can also be treated because the bacteria cannot thrive in high oxygen concentrations. In scuba diving (where scuba is an acronym formed from ‘self-contained under water breathing apparatus’), air is supplied at a higher pressure, so that the pressure within the diver’s chest matches the pressure exerted by the surrounding water. The latter increases by about 1 atm for each 10 m of descent. One unfortunate con sequence of breathing air at high pressures is that nitrogen is much more soluble in fatty tissues than in water, so it tends to dissolve in the central nervous system, bone marrow, and fat reserves. The result is nitrogen narcosis, with symptoms like intoxication. If the diver rises too rapidly to the surface, the nitrogen comes out of its lipid solution as bubbles, which causes the painful and sometimes fatal condition known as the bends. Many cases of scuba drowning appear to be consequences of arterial embolisms (obstructions in arteries caused by gas bubbles) and loss of consciousness as the air bubbles rise into the head.
 الاكثر قراءة في مواضيع عامة في الكيمياء الفيزيائية
الاكثر قراءة في مواضيع عامة في الكيمياء الفيزيائية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة


