


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 13-3-2019
Date: 25-11-2018
Date: 12-3-2017
|
Sulfur has an atomic weight of 32.066 grams per mole and is part of group 16, the oxygen family. It is a nonmetal and has a specific heat of 0.706 J g-1 oC-1. The electron affinity if 200 kJ mol-1 and the electronegativity is 2.58 (unitless). Sulfur is typically found as a light-yellow, opaque, and brittle solid in large amounts of small orthorhombic crystals. Not only does sulfur have twice the density of water, it is also insoluble in water. On the other hand, sulfur is highly soluble in carbon disulfide and slightly soluble in many common solvents. Sulfur can also vary in color and blackens upon boiling due to carbonaceous impurities. Even as little as 0.05% of carbonaceous matter darkens sulfur significantly.
Figure 1.1 : A piece of sulfur burning with a blue flame that can hardly be seen at daylight, but it can be seen in this photograph if you look closely. Image by Johannes Hemmerlein and used with permission.
Most sulfur is recovered directly as the element from underground deposits by injecting super-heated water and piping out molten sulfur (sulfur melts at 112o C). Compared to other elements, sulfur has the most allotropes. While the S8 ring is the most common allotrope, there are 6 other structures with up to 20 sulfur atoms per ring.
, S2, S4, S6, and S8
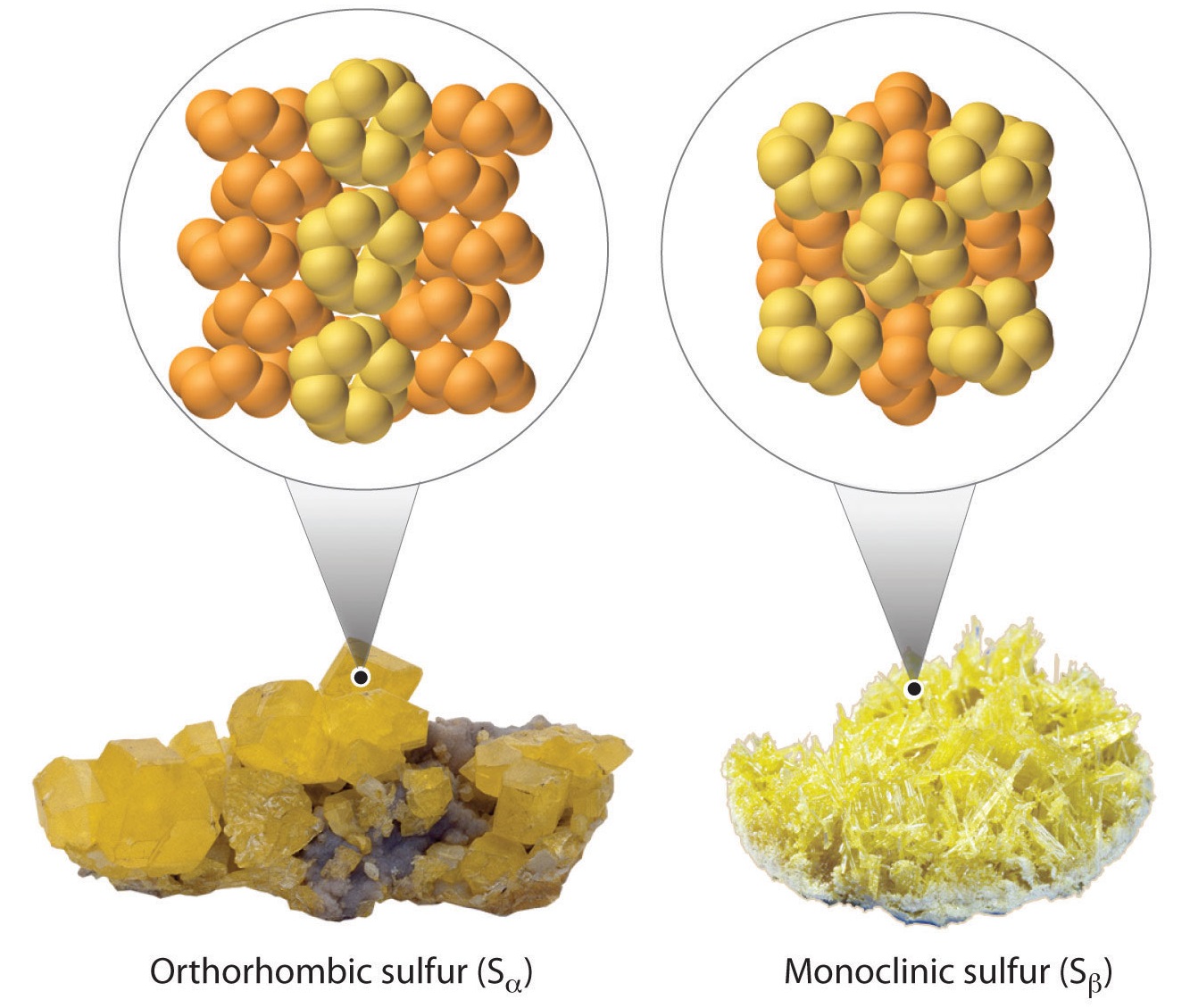
Figure 1.2 : Two allotropes of sulfur.
While oxygen has fewer allotropes than sulfur, including O , O2, O3, O4, O8, metallic O
(and four other solid phases), many of these actually have a corresponding sulfur variant. However, sulfur has more tendency to catenate (the linkage of atoms of the same element into longer chains). Here are the values of the single and double bond enthalpies:

This means that O=O is stronger than S=S, while O–O is weaker than S–S. So, in sulfur, single bonds are favored and catenation is easier than in oxygen compounds. It seems that the reason for the weaker S=S double bonds has its roots in the size of the atom: it's harder for the two atoms to come at a small enough distance, so that the p orbitals overlap is small and the π bond is weak. This is attested by looking down the periodic table: Se=Se has an even weaker bond enthalpy of 272kJ/mol .
What happens when the solid sulfur melts? The S8
molecules break up. When suddenly cooled, long chain molecules are formed in the plastic sulfur which behave as rubber. Plastic sulfur transform into rhombic sulfur over time.



|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
العتبة العباسية المقدسة تقدم دعوة إلى كلية مزايا الجامعة للمشاركة في حفل التخرج المركزي الخامس
|
|
|