


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 16-12-2019
Date: 16-12-2019
Date: 25-7-2018
|
Reactions of α-Amino Acids : Oxidative Coupling
The mild oxidant iodine reacts selectively with certain amino acid side groups. These include the phenolic ring in tyrosine, and the heterocyclic rings in tryptophan and histidine, which all yield products of electrophilic iodination. In addition, the sulfur groups in cysteine and methionine are also oxidized by iodine. Quantitative measurement of iodine consumption has been used to determine the number of such residues in peptides. The basic functions in lysine and arginine are onium cations at pH less than 8, and are unreactive in that state.
Cysteine is a thiol, and like most thiols it is oxidatively dimerized to a disulfide, which is sometimes listed as a distinct amino acid under the name cystine. Disulfide bonds of this kind are found in many peptides and proteins. For example, the two peptide chains that constitute insulin are held together by two disulfide links. Our hair consists of a fibrous protein called keratin, which contains an unusually large proportion of cysteine. In the manipulation called "permanent waving", disulfide bonds are first broken and then created after the hair has been reshaped. Treatment with dilute aqueous iodine oxidizes the methionine sulfur atom to a sulfoxide.
Cysteine-Cystine Interconversion
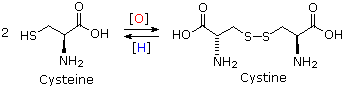



|
|
|
|
علامات بسيطة في جسدك قد تنذر بمرض "قاتل"
|
|
|
|
|
|
|
أول صور ثلاثية الأبعاد للغدة الزعترية البشرية
|
|
|
|
|
|
|
مدرسة دار العلم.. صرح علميّ متميز في كربلاء لنشر علوم أهل البيت (عليهم السلام)
|
|
|