


 النبات
النبات
 الحيوان
الحيوان
 الأحياء المجهرية
الأحياء المجهرية
 علم الأمراض
علم الأمراض
 التقانة الإحيائية
التقانة الإحيائية
 التقنية الحيوية المكروبية
التقنية الحيوية المكروبية
 التقنية الحياتية النانوية
التقنية الحياتية النانوية
 علم الأجنة
علم الأجنة
 الأحياء الجزيئي
الأحياء الجزيئي
 علم وظائف الأعضاء
علم وظائف الأعضاء
 الغدد
الغدد
 المضادات الحيوية
المضادات الحيوية|
Read More
Date: 3-12-2015
Date: 25-5-2021
Date: 1-5-2016
|
An LCR Can Control a Domain
KEY CONCEPTS
-Locus control regions are located at the 5′ end of a chromosomal domain and typically consist of multiple DNase hypersensitive sites.
-Locus control regions regulate gene clusters.
-Locus control regions usually regulate loci that show complex developmental or cell-type specific patterns of gene expression.
-Locus control regions control the transcription of target genes in the locus by direct interactions, forming looped structures.
Every gene is controlled by its proximal promoter, and most genes also respond to enhancers (containing similar regulatory elements located farther away; see the chapter titled Eukaryotic Transcription). These local controls are not sufficient for all genes, though. In some cases, a gene lies within a domain of several genes, all of which are influenced by specialized regulatory elements that act on the whole domain. The existence of these elements was identified by the inability of a region of DNA including a gene and all its known regulatory elements to be properly expressed when introduced into an animal as a transgene.
The best-characterized example of a regulated gene cluster is provided by the mammalian β-globin genes. Recall from the chapter titled Genome Sequences and Evolution that the α- and β- globin genes in mammals each exist as clusters of related genes that are expressed at different times and in different tissues during embryonic and adult development. These genes are associated with a large number of regulatory elements, which have been analyzed in detail. In the case of the adult human β-globin gene, regulatory sequences are located both 5′ and 3′ to the gene. The regulatory sequences include positive and negative elements in the promoter region as well as additional positive elements within and downstream of the gene.
All of these control regions are not, however, sufficient for proper expression of the human β-globin gene in a transgenic mouse within an order of magnitude of wild-type levels. Some further regulatory sequence is required. Regions that provide the additional regulatory function are identified by DNase I hypersensitive sites that are found at the ends of the β-globin cluster. The map in FIGURE 1 shows that the 20 kb upstream of the ε gene contains a group of 5 hypersensitive sites, and that there is a single site 30 kb downstream of the β gene.
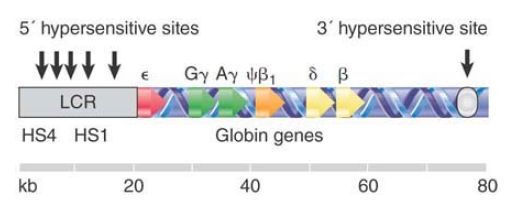
FIGURE 1. The β-globin locus is marked by hypersensitive sites at either end. The group of sites at the 5′ side constitutes the LCR and is essential for the function of all genes in the cluster.
The 5′ regulatory sites are the primary regulators, and the region containing the cluster of hypersensitive sites is called the locus control region (LCR). The role of the LCR is complex; in some ways it behaves as a “super enhancer” that poises the entire locus for transcription. The precise function of the 3′ hypersensitive site in the mammalian locus is not clear, but it is known to physically interact with the LCR. A 3′ hypersensitive site in the chicken β-globin locus acts as an insulator, as does a fifth 5′ site upstream of the mammalian LCR. The LCR is absolutely required for expression of each of the globin genes in the locus. Each gene is then further regulated by its own specific controls. Some of these controls are autonomous: Expression of the ε and γ genes appears intrinsic to those loci in conjunction with the LCR. Other controls appear to rely upon position in the cluster, which provides a suggestion that gene order in a cluster is important for regulation.
The entire region containing the globin genes, and extending well beyond them, constitutes a chromosomal domain. It shows increased sensitivity to digestion by DNase I. Deletion of the 5′
LCR restores normal resistance to DNase over the entire region. In addition to increases in the general accessibility of the locus, the LCR is also apparently required to directly activate the individual promoters. Researchers have not yet fully defined the exact nature of the sequential interactions between the LCR and the individual promoters, but it has recently become clear that the LCR contacts individual promoters directly, forming loops when these promoters are active. The domain controlled by the LCR also shows distinctive patterns of histone modifications that are dependent on LCR function.
This model appears to apply to other gene clusters, as well. The α- globin locus has a similar organization of genes that are expressed at different times, with a group of hypersensitive sites at one end of the cluster and increased sensitivity to DNase I throughout the region. So far, though, only a small number of other cases are known in which an LCR controls a group of genes.
One of these cases involves an LCR that controls genes on more than one chromosome. The TH2 LCR coordinately H regulates the T helper type 2 cytokine locus, a group of genes encoding a number of interleukins (important signaling molecules in the immune system). These genes are spread out over 120 kb on chromosome 11, and the T 2 LCR controls them by interacting with their promoters. It also interacts with the promoter of the IFNγ gene on chromosome 10. The two types of interaction are alternatives that comprise two different cell fates; that is, in one group of cells the LCR causes expression of the genes on chromosome 11, whereas in the other group it causes the gene on chromosome 10 to be expressed.
Looping interactions are important for chromosome structure, and function was introduced in the chapter titled Chromosomes. New methods have been developed to begin to dissect the physical interactions between chromosomal loci in vivo, leading to fresh understanding of how these interactions result in regulatory functions. Direct interactions between the β-globin and TH2 LCRs and their target loci have been mapped using a method known as chromosome conformation capture (3C). There are now many variations of this procedure; the basic method is outlined in the top panel of FIGURE 2. Interacting regions of chromatin in vivo are captured using formaldehyde treatment to crosslink to fix the DNA and proteins that are in close contact. Next, the chromatin is digested with a restriction enzyme and ligated under dilute conditions to favor intra-molecular ligation. This results in preferential ligation of DNA fragments that are held in close proximity as a result of crosslinking. Finally, the proteins are removed by reversing the crosslinking and the new ligated junctions are detected by PCR or sequencing.
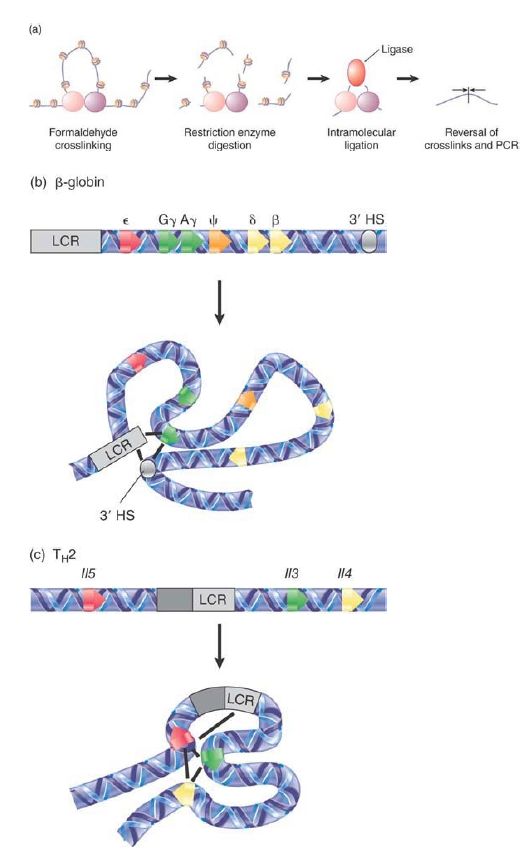
FIGURE 2. 3C is one method to detect physical interactions between regions of chromatin in vivo. Looping interactions controlled by the β-globin and TH2 LCRs have been mapped by 3C and some of the known contacts are shown.
Adapted from: Miele, A., and Dekker, J. 2008. Mol Biosyst 4:1046–1057.
As shown in the lower part of the Figure 8.51, 3C and similar methods have allowed researchers to begin to unravel the complex and dynamic interactions that occur at loci regulated by LCRs. The β-globin LCR sequentially interacts with each globin gene at the developmental stage in which that gene is active; the figure shows the interactions that occur between the LCR, 3′ HS, and the γ-globin genes in the fetal stage. Interestingly, the TH2 LCR appears to interact with all three of its target genes (Il3, −4, and −5) simultaneously. These interactions occur in all T-cells regardless of whether these genes are expressed, but the precise organization of loops alters upon activation of the interleukin genes. This reorganization, which depends on the protein SATB1 (special ATrich binding protein), suggests that the TH2 LCR brings all the genes together in a poised state in T cells, awaiting the trigger of specific transcription factors to activate the genes rapidly when needed.



|
|
|
|
تفوقت في الاختبار على الجميع.. فاكهة "خارقة" في عالم التغذية
|
|
|
|
|
|
|
أمين عام أوبك: النفط الخام والغاز الطبيعي "هبة من الله"
|
|
|
|
|
|
|
قسم شؤون المعارف ينظم دورة عن آليات عمل الفهارس الفنية للموسوعات والكتب لملاكاته
|
|
|