


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 30-10-2018
Date: 29-11-2018
Date: 11-10-2018
|
The hydride ion
The enthalpy change ΔEAH(298 K) associated with the attachment of an electron to an H atom (reaction 1.1) is ≈73 kJ mol_1.
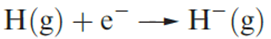 (1.1)
(1.1)
All alkali metal hydrides crystallize with the NaCl lattice. From diffraction data and the ionic radii of the metal ions (Appendix 6) the radius of H_ can be estimated using equation 1.2; it varies from 130pm (in LiH) to 154pm (in CsH) and can be considered similar to that of F- (133 pm).
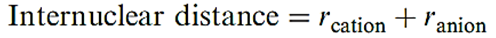 (1.2)
(1.2)
The large increase in size on going from the H atom (rcov = 37 pm) to the H- ion arises from interelectronic repulsion when a second electron enters the 1s atomic orbital. The smaller rH- in LiH may suggest some degree of covalent bonding, but calculated and experimental values of lattice energies for each of the group 1 metal hydrides are in good agreement,suggesting that an electrostatic model is appropriate for each compound. Hydrides of the s-block metals (excluding Be) can be made by heating the metal with H2.
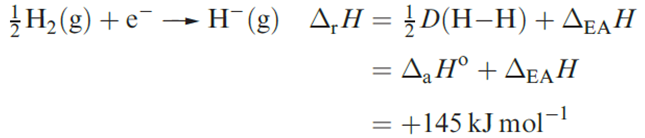 (1.3)
(1.3)
When we compare ΔrH for reaction 1.3 with those for the formations of F_ and Cl- from F2 and Cl2 (_249 and _228 kJ mol-1, respectively), we understand why, since H_ is about the same size as F-, ionic hydrides are relatively unstable species with respect to dissociation into their constituent elements. Salt-like hydrides of metals in high oxidation states are most unlikely to exist.



|
|
|
|
علامات بسيطة في جسدك قد تنذر بمرض "قاتل"
|
|
|
|
|
|
|
أول صور ثلاثية الأبعاد للغدة الزعترية البشرية
|
|
|
|
|
|
|
معهد الكفيل للنطق والتأهيل: أطلقنا برامج متنوعة لدعم الأطفال وتعزيز مهاراتهم التعليمية والاجتماعية
|
|
|