
آخر المواضيع المضافة


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 20-5-2020
Date: 16-2-2018
Date: 5-5-2019
|
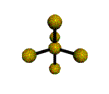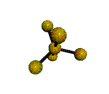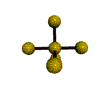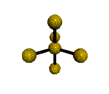 |
AX5 |
 |
Shape: | trigonal bipyramidal |
| Steric Number: | 5 | |
| Lone Pairs: | 0 | |
| Polar/NonPolar: | NonPolar | |
| Hybridization: | sp3d | |
| Examples: | PCl5, SbF5 |
NOTES: This molecule is made up of 5 sp3d hybrid orbitals. Three orbitals are arranged around the equator of the molecule with bond angles of 120o. Two orbitals are arranged along the vertical axis at 90o from the equatorial orbitals. The shape of the orbitals is trigonal bipyramidal. Since there is an atom at the end of each orbital, the shape of the molecule is also trigonal bipyramidal.



|
|
|
|
دراسة يابانية لتقليل مخاطر أمراض المواليد منخفضي الوزن
|
|
|
|
|
|
|
اكتشاف أكبر مرجان في العالم قبالة سواحل جزر سليمان
|
|
|
|
|
|
|
المجمع العلمي ينظّم ندوة حوارية حول مفهوم العولمة الرقمية في بابل
|
|
|