
آخر المواضيع المضافة


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 13-11-2020
Date: 3-1-2017
Date: 22-3-2017
|
Bidentate ligands have two lone pairs, both of which can bond to the central metal ion. The two commonly used examples are 1,2-diaminoethane (old name: ethylenediamine - often given the abbreviation "en"), and the ethanedioate ion (old name: oxalate).
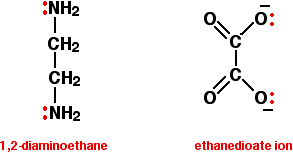
In the ethanedioate ion, there are lots more lone pairs than the two shown, but these are the only ones we are interested in. You can think of these bidentate ligands rather as if they were a pair of headphones, carrying lone pairs on each of the "ear pieces". These will then fit snuggly around a metal ion.
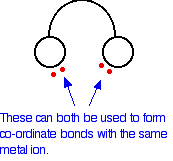



|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
قسم شؤون المعارف ووفد من جامعة البصرة يبحثان سبل تعزيز التعاون المشترك
|
|
|