
Electron density and the electrostatic potential surfaces
 المؤلف:
Peter Atkins، Julio de Paula
المؤلف:
Peter Atkins، Julio de Paula
 المصدر:
ATKINS PHYSICAL CHEMISTRY
المصدر:
ATKINS PHYSICAL CHEMISTRY
 الجزء والصفحة:
396
الجزء والصفحة:
396
 2025-11-30
2025-11-30
 9
9
Electron density and the electrostatic potential surfaces
One of the most significant developments in computational chemistry has been the introduction of graphical representations of molecular orbitals and electron densities. The raw output of a molecular structure calculation is a list of the coefficients of the atomic orbitals in each molecular orbital and the energies of these orbitals. The graphical representation of a molecular orbital uses stylized shapes to represent the basis set, and then scales their size to indicate the coefficient in the linear combination. Different signs of the wavefunctions are represented by different colours. Once the coefficients are known, we can build up a representation of the electron density in the molecule by noting which orbitals are occupied and then forming the squares of those orbitals. The total electron density at any point is then the sum of the squares of the wavefunctions evaluated at that point. The outcome is commonly represented by a isodensity surface, a surface of constant total electron density (Fig. 11.43). As shown in the illustration, there are several styles of representing an isodensity sur face, as a solid form, as a transparent form with a ball-and-stick representation of the molecule within, or as a mesh. A related representation is a solvent-accessible surface in which the shape represents the shape of the molecule by imagining a sphere representing a solvent molecule rolling across the surface and plotting the locations of the centre of that sphere. One of the most important aspects of a molecule other than its geometrical shape is the distribution of charge over its surface. The net charge at each point on an isodensity surface can be calculated by subtracting the charge due to the electron density at that point form the charge due to the nuclei: the result is an electrostatic potential sur face (an ‘elpot-surface’) in which net positive charge is shown in one colour and net negative charge is shown in another, with intermediate gradations of colour (Fig. 11.44). Representations such as those we have illustrated are of critical importance in a number of fields. For instance, they may be used to identify an electron-poor region of a molecule that is susceptible to association with or chemical attack by an electron rich region of another molecule. Such considerations are important for assessing the pharmacological activity of potential drugs.
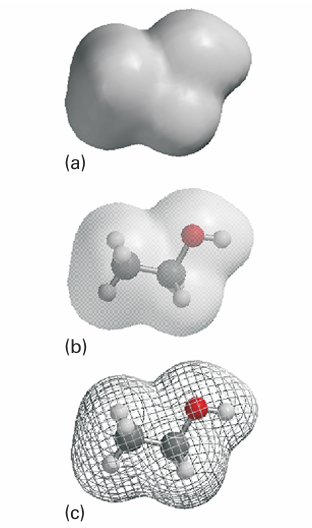
Fig. 11.43 Various representations of an isodensity surface of ethanol (a) solid surface, (b) transparent surface, and (c) mesh surface.

Fig. 11.44 An elpot diagram of ethanol.
 الاكثر قراءة في مواضيع عامة في الكيمياء الفيزيائية
الاكثر قراءة في مواضيع عامة في الكيمياء الفيزيائية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة


