
Benzene and aromatic stability
 المؤلف:
Peter Atkins، Julio de Paula
المؤلف:
Peter Atkins، Julio de Paula
 المصدر:
ATKINS PHYSICAL CHEMISTRY
المصدر:
ATKINS PHYSICAL CHEMISTRY
 الجزء والصفحة:
ص391-392
الجزء والصفحة:
ص391-392
 2025-11-30
2025-11-30
 8
8
Benzene and aromatic stability
The most notable example of delocalization conferring extra stability is benzene and the aromatic molecules based on its structure. Benzene is often expressed in a mixture of valence-bond and molecular orbital terms, with typically valence-bond language used for its σ framework and molecular orbital language used to describe its π electrons. First, the valence-bond component. The six C atoms are regarded as sp2hybridized, with a single unhydridized perpendicular 2porbital. One H atom is bonded by (Csp2, H1s) overlap to each C carbon, and the remaining hybrids overlap to give a regular hexagon of atoms (Fig. 11.40). The internal angle of a regular hexagon is 120°, sosp2hybridization is ideally suited for forming σ bonds. We see that benzene’s hexagonal shape permits strain-free σ bonding. Now consider the molecular orbital component of the description. The six C2p orbitals overlap to give six πorbitals that spread all round the ring. Their energies are calculated within the Hückel approximation by diagonalizing the hamiltonian matrix

The MO energies, the eigenvalues of this matrix, are simply E=α±2β, α±β, α±β (11.49) as shown in Fig. 11.41. The orbitals there have been given symmetry labels that we explain in Chapter 12. Note that the lowest energy orbital is bonding between all neighbouring atoms, the highest energy orbital is antibonding between each pair of neighbours, and the intermediate orbitals are a mixture of bonding, nonbonding, and antibonding character between adjacent atoms.
We now apply the building-up principle to the π system. There are six electrons to accommodate (one from each C atom), so the three lowest orbitals (a2u and the doubly degenerate pair e1g) are fully occupied, giving the ground-state configuration a22ue41g. A significant point is that the only molecular orbitals occupied are those with net bonding character. Theπ-electron energy of benzene is
Eπ =2(α+2β) +4(α+β) =6α+8β
If we ignored delocalization and thought of the molecule as having three isolated π bonds, it would be ascribed a π-electron energy of only 3(2α + 2β) = 6α + 6β. The delocalization energy is therefore 2β ≈−460 kJ mol−1, which is considerably more than for butadiene. The π-bond formation energy in benzene is 8β. This discussion suggests that aromatic stability can be traced to two main contributions. First, the shape of the regular hexagon is ideal for the formation of strong σ bonds: the σ framework is relaxed and without strain. Second, the π orbitals are such as to be able to accommodate all the electrons in bonding orbitals, and the delocalization energy is large.
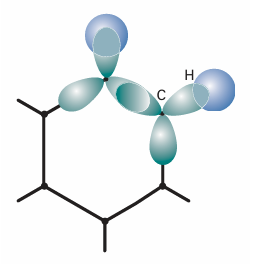
Fig. 11.40Theσframework of benzene is formed by the overlap of Csp2hybrids, which fit without strain into a hexagonal arrangement.
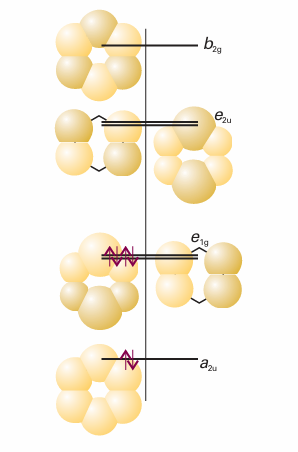
Fig. 11.41The Hückel orbitals of benzene and the corresponding energy levels. The symmetry labels are explained in Chapter 12. The bonding and antibonding character of the delocalized orbitals reflects the numbers of nodes between the atoms. In the ground state, only the bonding orbitals are occupied.
 الاكثر قراءة في مواضيع عامة في الكيمياء الفيزيائية
الاكثر قراءة في مواضيع عامة في الكيمياء الفيزيائية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة


