


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 24-8-2016
Date: 19-5-2019
Date: 8-8-2016
|
Sometimes it is inconvenient to use the concentration units during a reaction a species’ concentration may change by many orders of magnitude. If we want to display the reaction’s progress graphically we might plot the reactant’s concentration as a function of time or as a function of the volume of a reagent being added to the reaction. Such is the case in Figure 1.1 for the titration of HCl with NaOH.
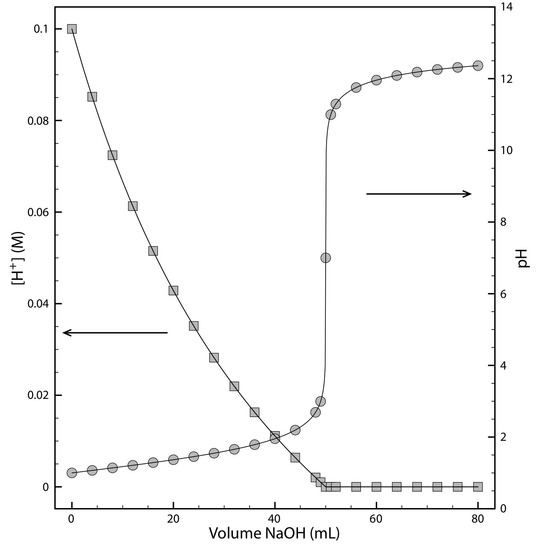
Figure 1.1: Graph showing the progress for the titration of 50.0 mL of 0.10 M HCl with 0.10 M NaOH. The [H+] is shown on the left y-axis and the pH on the right y-axis.
The y-axis on the left-side of the figure displays the [H+] as a function of the volume of NaOH. The initial [H+] is 0.10 M and its concentration after adding 80 mL of NaOH is 4.3 × 10-13 M. We can easily follow the change in [H+] for the first 14 additions of NaOH. For the remaining additions of NaOH, however, the change in [H+] is too small to see.
When working with concentrations spanning many orders of magnitude, it is often more convenient to express concentration using a p-function. The p-function of X is written as pX and is defined as
The pH of a solution that is 0.10 M H+ is
and the pH of 4.3 × 10-13 M H+ is



|
|
|
|
التوتر والسرطان.. علماء يحذرون من "صلة خطيرة"
|
|
|
|
|
|
|
مرآة السيارة: مدى دقة عكسها للصورة الصحيحة
|
|
|
|
|
|
|
نحو شراكة وطنية متكاملة.. الأمين العام للعتبة الحسينية يبحث مع وكيل وزارة الخارجية آفاق التعاون المؤسسي
|
|
|