


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 30-9-2019
Date: 8-9-2020
Date: 7-10-2020
|
If the electron pairs in covalent bonds were donated and shared absolutely evenly there would be no fixed local charges within a molecule. Although this is true for diatomic elements such as H2, N2 and O2, most covalent compounds show some degree of local charge separation, resulting in bond and / or molecular dipoles. A dipole exists when the centers of positive and negative charge distribution do not coincide.
A large local charge separation usually results when a shared electron pair is donated unilaterally. The three Kekulé formulas shown here illustrate this condition.
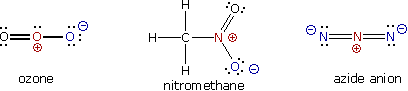
In the formula for ozone the central oxygen atom has three bonds and a full positive charge while the right hand oxygen has a single bond and is negatively charged. The overall charge of the ozone molecule is therefore zero. Similarly, nitromethane has a positive-charged nitrogen and a negative-charged oxygen, the total molecular charge again being zero. Finally, azide anion has two negative-charged nitrogens and one positive-charged nitrogen, the total charge being minus one.
In general, for covalently bonded atoms having valence shell electron octets, if the number of covalent bonds to an atom is greater than its normal valence it will carry a positive charge. If the number of covalent bonds to an atom is less than its normal valence it will carry a negative charge. The formal charge on an atom may also be calculated by the following formula:
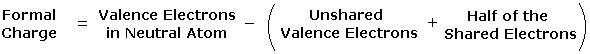



|
|
|
|
للعاملين في الليل.. حيلة صحية تجنبكم خطر هذا النوع من العمل
|
|
|
|
|
|
|
"ناسا" تحتفي برائد الفضاء السوفياتي يوري غاغارين
|
|
|
|
|
|
|
بالفيديو والصور: العتبة الحسينية تواصل دعم القطاع الزراعي عبر مدينة سيد الشهداء (ع) الزراعية
|
|
|