

علم الكيمياء

تاريخ الكيمياء والعلماء المشاهير

التحاضير والتجارب الكيميائية

المخاطر والوقاية في الكيمياء

اخرى

مقالات متنوعة في علم الكيمياء

كيمياء عامة


الكيمياء التحليلية

مواضيع عامة في الكيمياء التحليلية

التحليل النوعي والكمي

التحليل الآلي (الطيفي)

طرق الفصل والتنقية


الكيمياء الحياتية

مواضيع عامة في الكيمياء الحياتية

الكاربوهيدرات

الاحماض الامينية والبروتينات

الانزيمات

الدهون

الاحماض النووية

الفيتامينات والمرافقات الانزيمية

الهرمونات


الكيمياء العضوية

مواضيع عامة في الكيمياء العضوية

الهايدروكاربونات

المركبات الوسطية وميكانيكيات التفاعلات العضوية

التشخيص العضوي

تجارب وتفاعلات في الكيمياء العضوية


الكيمياء الفيزيائية

مواضيع عامة في الكيمياء الفيزيائية

الكيمياء الحرارية

حركية التفاعلات الكيميائية

الكيمياء الكهربائية


الكيمياء اللاعضوية

مواضيع عامة في الكيمياء اللاعضوية

الجدول الدوري وخواص العناصر

نظريات التآصر الكيميائي

كيمياء العناصر الانتقالية ومركباتها المعقدة


مواضيع اخرى في الكيمياء

كيمياء النانو

الكيمياء السريرية

الكيمياء الطبية والدوائية

كيمياء الاغذية والنواتج الطبيعية

الكيمياء الجنائية


الكيمياء الصناعية

البترو كيمياويات

الكيمياء الخضراء

كيمياء البيئة

كيمياء البوليمرات

مواضيع عامة في الكيمياء الصناعية

الكيمياء التناسقية

الكيمياء الاشعاعية والنووية
Anionic Polymerization
المؤلف:
sami matar & Lewis. F. Hatch
المصدر:
Chemistry of PETROCHEMICAL PROCESSES
الجزء والصفحة:
p 308
18-9-2017
1896
Anionic Polymerization
Anionic polymerization is better for vinyl monomers with electron withdrawing groups that stabilize the intermediates. Typical monomers best polymerized by anionic initiators include acrylonitrile, styrene, and butadiene. As with cationic polymerization, a counter ion is present with the propagating chain. The propagation and the termination steps are similar to cationic polymerization.
Many initiators, such as alkyl and aryllithium and sodium and lithium suspensions in liquid ammonia, effect the polymerization. For example, acrylonitrile combined with n-butyllithium forms a carbanion intermediate:

Chain growth occurs through a nucleophilic attack of the carbanion on the monomer. As in cationic polymerizations, lower temperatures favor anionic polymerizations by minimizing branching due to chain transfer reactions.
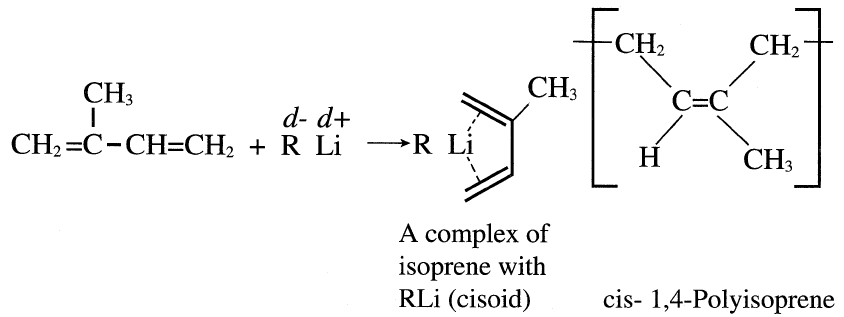
Solvent polarity is also important in directing the reaction bath and the composition and orientation of the products. For example, the polymerization of butadiene with lithium in tetrahydrofuran (a polar solvent) gives a high 1,2 addition polymer. Polymerization of either butadiene or isoprene using lithium compounds in nonpolar solvent such as n-pentane produces a high cis-1,4 addition product. However, a higher cis-1,4-polyisoprene isomer was obtained than when butadiene was used. This occurs because butadiene exists mainly in a transoid conformation at room temperature (a higher cisoid conformation is anticipated for isoprene):
 الاكثر قراءة في البترو كيمياويات
الاكثر قراءة في البترو كيمياويات
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















