

علم الكيمياء

تاريخ الكيمياء والعلماء المشاهير

التحاضير والتجارب الكيميائية

المخاطر والوقاية في الكيمياء

اخرى

مقالات متنوعة في علم الكيمياء

كيمياء عامة


الكيمياء التحليلية

مواضيع عامة في الكيمياء التحليلية

التحليل النوعي والكمي

التحليل الآلي (الطيفي)

طرق الفصل والتنقية


الكيمياء الحياتية

مواضيع عامة في الكيمياء الحياتية

الكاربوهيدرات

الاحماض الامينية والبروتينات

الانزيمات

الدهون

الاحماض النووية

الفيتامينات والمرافقات الانزيمية

الهرمونات


الكيمياء العضوية

مواضيع عامة في الكيمياء العضوية

الهايدروكاربونات

المركبات الوسطية وميكانيكيات التفاعلات العضوية

التشخيص العضوي

تجارب وتفاعلات في الكيمياء العضوية


الكيمياء الفيزيائية

مواضيع عامة في الكيمياء الفيزيائية

الكيمياء الحرارية

حركية التفاعلات الكيميائية

الكيمياء الكهربائية


الكيمياء اللاعضوية

مواضيع عامة في الكيمياء اللاعضوية

الجدول الدوري وخواص العناصر

نظريات التآصر الكيميائي

كيمياء العناصر الانتقالية ومركباتها المعقدة


مواضيع اخرى في الكيمياء

كيمياء النانو

الكيمياء السريرية

الكيمياء الطبية والدوائية

كيمياء الاغذية والنواتج الطبيعية

الكيمياء الجنائية


الكيمياء الصناعية

البترو كيمياويات

الكيمياء الخضراء

كيمياء البيئة

كيمياء البوليمرات

مواضيع عامة في الكيمياء الصناعية

الكيمياء التناسقية

الكيمياء الاشعاعية والنووية
Relativistic effects of The group 13 elements
المؤلف:
CATHERINE E. HOUSECROFT AND ALAN G. SHARPE
المصدر:
Inorganic Chemistry
الجزء والصفحة:
p 298
24-1-2018
2148
Relativistic effects of The group 13 elements
Among many generalizations about heavier elements are two that depend on quantum theory for explanation:
- the ionization energies of the 6s electrons are anomalously high, leading to the marked stabilization of Hg(0), Tl(I), Pb(II) and Bi(III) compared with Cd(0), In(I), Sn(II) and Sb(III);
- whereas bond energies usually decrease down a group of p-block elements, they often increase down a group of d-block metals, in both the elements themselves and their compounds.
These observations can be accounted for (though often far from simply) if Einstein’s theory of relativity is combined with quantum mechanics, in which case they are attributed to relativistic effects. We focus here on chemical generalizations. According to the theory of relativity, the mass m of a particle increases from its rest mass m0 when its velocity v approaches the speed of light, c, and m is then given by the equation:
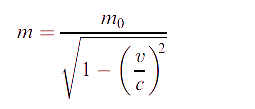
For a one-electron system, the Bohr model of the atom (which, despite its shortcomings, gives the correct value for the ionization energy) leads to the velocity of the electron being expressed by the equation:

the increase in m results in an approximately 20% contraction of the radius of the 1s (n = 1) orbital; this is called relativistic contraction. Other s orbitals are affected in a similar way and as a consequence, when Z is high, s orbitals have diminished overlap with orbitals of other atoms. A detailed treatment shows that p orbitals (which have a low electron density near to the nucleus) are less affected. On the other hand, d orbitals (which are more effectively screened from the nuclear charge by the contracted s and p orbitals) undergo a relativistic expansion; a similar argument applies to f orbitals.
 الاكثر قراءة في مواضيع عامة في الكيمياء اللاعضوية
الاكثر قراءة في مواضيع عامة في الكيمياء اللاعضوية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















