
تاريخ الفيزياء

علماء الفيزياء


الفيزياء الكلاسيكية

الميكانيك

الديناميكا الحرارية


الكهربائية والمغناطيسية

الكهربائية

المغناطيسية

الكهرومغناطيسية


علم البصريات

تاريخ علم البصريات

الضوء

مواضيع عامة في علم البصريات

الصوت


الفيزياء الحديثة


النظرية النسبية

النظرية النسبية الخاصة

النظرية النسبية العامة

مواضيع عامة في النظرية النسبية

ميكانيكا الكم

الفيزياء الذرية

الفيزياء الجزيئية


الفيزياء النووية

مواضيع عامة في الفيزياء النووية

النشاط الاشعاعي


فيزياء الحالة الصلبة

الموصلات

أشباه الموصلات

العوازل

مواضيع عامة في الفيزياء الصلبة

فيزياء الجوامد


الليزر

أنواع الليزر

بعض تطبيقات الليزر

مواضيع عامة في الليزر


علم الفلك

تاريخ وعلماء علم الفلك

الثقوب السوداء


المجموعة الشمسية

الشمس

كوكب عطارد

كوكب الزهرة

كوكب الأرض

كوكب المريخ

كوكب المشتري

كوكب زحل

كوكب أورانوس

كوكب نبتون

كوكب بلوتو

القمر

كواكب ومواضيع اخرى

مواضيع عامة في علم الفلك

النجوم

البلازما

الألكترونيات

خواص المادة


الطاقة البديلة

الطاقة الشمسية

مواضيع عامة في الطاقة البديلة

المد والجزر

فيزياء الجسيمات


الفيزياء والعلوم الأخرى

الفيزياء الكيميائية

الفيزياء الرياضية

الفيزياء الحيوية

الفيزياء والفلسفة

الفيزياء العامة


مواضيع عامة في الفيزياء

تجارب فيزيائية

مصطلحات وتعاريف فيزيائية

وحدات القياس الفيزيائية

طرائف الفيزياء

مواضيع اخرى
Transient Radioactive Equilibrium
المؤلف:
U.S. Department of Commerce, National Technical Information Service, 1993
المصدر:
The Nuclear Physics and Reactor Theory Handbook
الجزء والصفحة:
p 40
29-3-2017
2565
Transient Radioactive Equilibrium
Transient radioactive equilibrium occurs when the parent nuclide and the daughter nuclide decay at essentially the same rate.
For transient equilibrium to occur, the parent must have a long half-life when compared to the daughter. An example of this type of compound decay process is barium-140, which decays by beta emission to lanthanum-140, which in turn decays by beta emission to stable cerium-140.
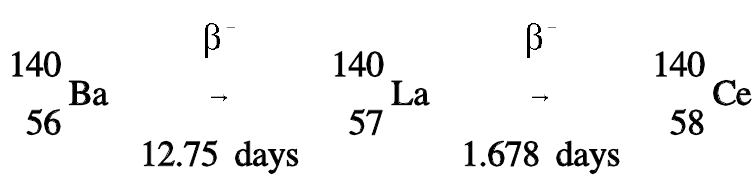
The decay constant for barium-140 is considerably smaller than the decay constant for lanthanum-140. Remember that the rate of decay of both the parent and daughter can be represented as λN. Although the decay constant for barium-140 is smaller, the actual rate of decay (λN) is initially larger than that of lanthanum-140 because of the great difference in their initial concentrations. As the concentration of the daughter increases, the rate of decay of the daughter will approach and eventually match the decay rate of the parent. When this occurs, they are said to be in transient equilibrium. A plot of the barium-lanthanum-cerium decay chain reaching transient equilibrium is shown in Figure 1.
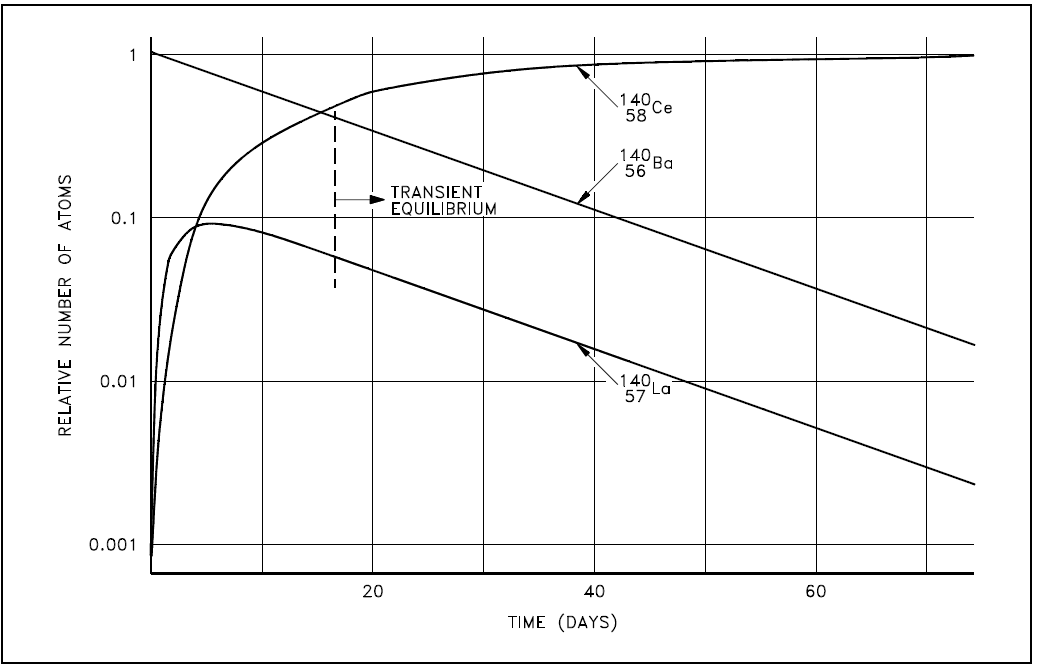
Figure 1: Transient Equilibrium in the Decay of Barium-140
Secular equilibrium occurs when the parent has an extremely long half-life. In the long decay chain for a naturally radioactive element, such as thorium-232, where all of the elements in the chain are in secular equilibrium, each of the descendants has built up to an equilibrium amount and all decay at the rate set by the original parent. The only exception is the final stable element on the end of the chain. Its number of atoms is constantly increasing.
 الاكثر قراءة في النشاط الاشعاعي
الاكثر قراءة في النشاط الاشعاعي
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















