
Atomic and Nuclear Radii
 المؤلف:
U.S. Department of Commerce, National Technical Information Service, 1993
المؤلف:
U.S. Department of Commerce, National Technical Information Service, 1993
 المصدر:
The Nuclear Physics and Reactor Theory Handbook
المصدر:
The Nuclear Physics and Reactor Theory Handbook
 الجزء والصفحة:
p 6
الجزء والصفحة:
p 6
 25-3-2017
25-3-2017
 2507
2507
Atomic and Nuclear Radii
The size of an atom is difficult to define exactly due to the fact that the electron cloud, formed by the electrons moving in their various orbitals, does not have a distinct outer edge. A reasonable measure of atomic size is given by the average distance of the outermost electron from the nucleus. Except for a few of the lightest atoms, the average atomic radii are approximately the same for all atoms, about 2 x 10 -8 cm.
Like the atom the nucleus does not have a sharp outer boundary. Experiments have shown that the nucleus is shaped like a sphere with a radius that depends on the atomic mass number of the atom. The relationship between the atomic mass number and the radius of the nucleus is shown in the following equation.
r = (1.25 x 10 -13 cm) A1/3
where:
r = radius of the nucleus (cm)
A = atomic mass number (dimensionless)
The values of the nuclear radii for some light, intermediate, and heavy nuclides are shown in Table 1.
TABLE 1: Calculated Values for Nuclear Radii
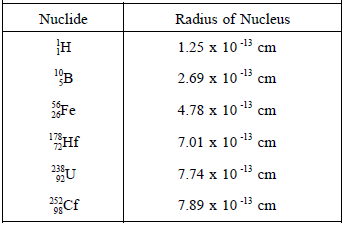
From the table, it is clear that the radius of a typical atom (e.g. 2 x 10 -8 cm) is more than 25,000 times larger than the radius of the largest nucleus.
 الاكثر قراءة في مواضيع عامة في الفيزياء النووية
الاكثر قراءة في مواضيع عامة في الفيزياء النووية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة


