

علم الكيمياء

تاريخ الكيمياء والعلماء المشاهير

التحاضير والتجارب الكيميائية

المخاطر والوقاية في الكيمياء

اخرى

مقالات متنوعة في علم الكيمياء

كيمياء عامة


الكيمياء التحليلية

مواضيع عامة في الكيمياء التحليلية

التحليل النوعي والكمي

التحليل الآلي (الطيفي)

طرق الفصل والتنقية


الكيمياء الحياتية

مواضيع عامة في الكيمياء الحياتية

الكاربوهيدرات

الاحماض الامينية والبروتينات

الانزيمات

الدهون

الاحماض النووية

الفيتامينات والمرافقات الانزيمية

الهرمونات


الكيمياء العضوية

مواضيع عامة في الكيمياء العضوية

الهايدروكاربونات

المركبات الوسطية وميكانيكيات التفاعلات العضوية

التشخيص العضوي

تجارب وتفاعلات في الكيمياء العضوية


الكيمياء الفيزيائية

مواضيع عامة في الكيمياء الفيزيائية

الكيمياء الحرارية

حركية التفاعلات الكيميائية

الكيمياء الكهربائية


الكيمياء اللاعضوية

مواضيع عامة في الكيمياء اللاعضوية

الجدول الدوري وخواص العناصر

نظريات التآصر الكيميائي

كيمياء العناصر الانتقالية ومركباتها المعقدة


مواضيع اخرى في الكيمياء

كيمياء النانو

الكيمياء السريرية

الكيمياء الطبية والدوائية

كيمياء الاغذية والنواتج الطبيعية

الكيمياء الجنائية


الكيمياء الصناعية

البترو كيمياويات

الكيمياء الخضراء

كيمياء البيئة

كيمياء البوليمرات

مواضيع عامة في الكيمياء الصناعية

الكيمياء التناسقية

الكيمياء الاشعاعية والنووية
Coordination number 8
المؤلف:
CATHERINE E. HOUSECROFT AND ALAN G. SHARPE
المصدر:
INORGANIC CHEMISTRY
الجزء والصفحة:
2th ed p 546
27-2-2017
2026
Coordination number 8
As the number of vertices in a polyhedron increases, so does the number of possible structures (Figure 1.1a). Probably, the best known eight-vertex polyhedron is the cube, (1.1), but this is hardly ever observed as an arrangement of donor atoms in complexes. The few examples include the anions in the actinoid complexes Na3[PaF8], Na3[UF8] and [Et4N]4[U(NCS-N)8]. Steric hindrance between ligands can be reduced by converting a cubic into a square antiprismatic arrangement, i.e. on going from 1.1 to 1.2.
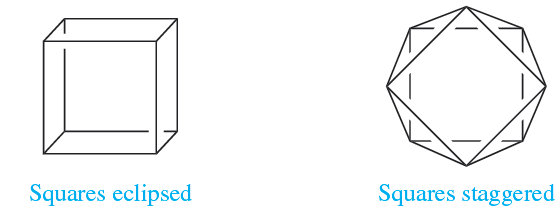
(1.1) (1.2)
Square antiprismatic coordination environments occur in [Zr(acac)4] (d0) and in the anions in the salts Na3[TaF8] (d0), K2[ReF8] (d1) and K2[H3NCH2CH2NH3][Nb)ox(4] (d1) (Figure 1.1b). Specifying the counter-ion is important since the energy difference between 8-coordinate structures tends to be small with the result that the preference between two structures may be altered by crystal packing forces in two different salts. Examples are seen in a range of salts of [Mo)CN(8]3- , [W(CN)8]3- , [Mo(CN)8]4- or [W(CN)8]4- which possess square antiprismatic or dodecahedral structures depending on the cation. Further examples of dodecahedral complexes include [Y(H2O)8]3+ (Figure 1.1c) and a number of complexes with didentate ligands: [Mo(O2(4]2- (d0), [Ti(NO3)4] (d0), [Cr(O2(4]3- (d1), [Mn(NO3)4]2- (d5) and [Fe(NO3)4]- (d5).
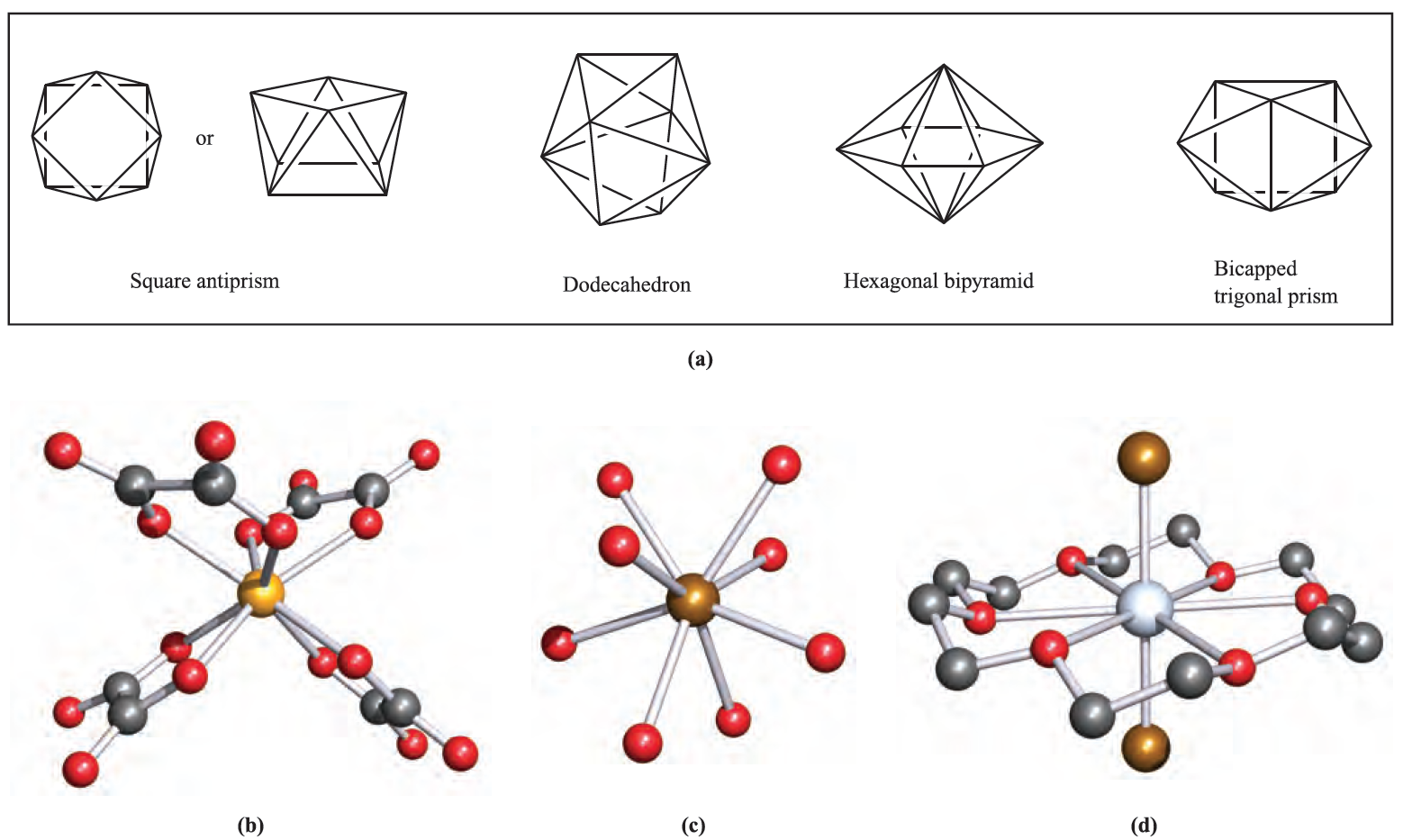
Fig. 1.1 (a) The coordination spheres defined by the donor atoms in idealized 8-coordinate structures; the left-hand drawing of the square antiprism emphasizes that the two square faces are mutually staggered. Examples of 8-coordinate complexes (X-ray diffraction): (b) the square antiprismatic structure of [Nb(ox)4]4- in the salt K2[H3NCH2CH2NH3][Nb(ox)4].4H2O [F.A. Cotton et al. (1987) Inorg. Chem., vol. 26, p. 2889]; (c) the dodecahedral ion [Y)H2O(8]3+ in the salt [Y(H2O)8]Cl3.(15-crown-5)[R.D. Rogers et al. (1986) Inorg. Chim. Acta, vol. 116, p. 171]; and (d) [CdBr2(18-crown-6(] with the macrocyclic ligand occupying the equatorial plane of a hexagonal bipyramid [A. Hazell (1988) Acta Crystallogr., Sect. C, vol. 44, p. 88]. Hydrogen atoms have been omitted for clarity; colour code: Nb, yellow; O, red; Y, brown; Cd, silver; C, grey; Br, brown.
The hexagonal bipyramid is a rare coordination environment, but may be favoured in complexes containing a hexadentate macrocyclic ligand, for example [CdBr2(18- crown-6)], Figure 1.1d. A bicapped trigonal prism is another option for 8-coordination, but is only rarely observed, e.g. in [ZrF8]4- (d0) and [La(acac)3(H2O)2].H2O (d0).
 الاكثر قراءة في كيمياء العناصر الانتقالية ومركباتها المعقدة
الاكثر قراءة في كيمياء العناصر الانتقالية ومركباتها المعقدة
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















