

النبات

مواضيع عامة في علم النبات

الجذور - السيقان - الأوراق

النباتات الوعائية واللاوعائية

البذور (مغطاة البذور - عاريات البذور)

الطحالب

النباتات الطبية


الحيوان

مواضيع عامة في علم الحيوان

علم التشريح

التنوع الإحيائي

البايلوجيا الخلوية


الأحياء المجهرية

البكتيريا

الفطريات

الطفيليات

الفايروسات


علم الأمراض

الاورام

الامراض الوراثية

الامراض المناعية

الامراض المدارية

اضطرابات الدورة الدموية

مواضيع عامة في علم الامراض

الحشرات


التقانة الإحيائية

مواضيع عامة في التقانة الإحيائية


التقنية الحيوية المكروبية

التقنية الحيوية والميكروبات

الفعاليات الحيوية

وراثة الاحياء المجهرية

تصنيف الاحياء المجهرية

الاحياء المجهرية في الطبيعة

أيض الاجهاد

التقنية الحيوية والبيئة

التقنية الحيوية والطب

التقنية الحيوية والزراعة

التقنية الحيوية والصناعة

التقنية الحيوية والطاقة

البحار والطحالب الصغيرة

عزل البروتين

هندسة الجينات


التقنية الحياتية النانوية

مفاهيم التقنية الحيوية النانوية

التراكيب النانوية والمجاهر المستخدمة في رؤيتها

تصنيع وتخليق المواد النانوية

تطبيقات التقنية النانوية والحيوية النانوية

الرقائق والمتحسسات الحيوية

المصفوفات المجهرية وحاسوب الدنا

اللقاحات

البيئة والتلوث


علم الأجنة

اعضاء التكاثر وتشكل الاعراس

الاخصاب

التشطر

العصيبة وتشكل الجسيدات

تشكل اللواحق الجنينية

تكون المعيدة وظهور الطبقات الجنينية

مقدمة لعلم الاجنة


الأحياء الجزيئي

مواضيع عامة في الاحياء الجزيئي


علم وظائف الأعضاء


الغدد

مواضيع عامة في الغدد

الغدد الصم و هرموناتها

الجسم تحت السريري

الغدة النخامية

الغدة الكظرية

الغدة التناسلية

الغدة الدرقية والجار الدرقية

الغدة البنكرياسية

الغدة الصنوبرية

مواضيع عامة في علم وظائف الاعضاء

الخلية الحيوانية

الجهاز العصبي

أعضاء الحس

الجهاز العضلي

السوائل الجسمية

الجهاز الدوري والليمف

الجهاز التنفسي

الجهاز الهضمي

الجهاز البولي


المضادات الميكروبية

مواضيع عامة في المضادات الميكروبية

مضادات البكتيريا

مضادات الفطريات

مضادات الطفيليات

مضادات الفايروسات

علم الخلية

الوراثة

الأحياء العامة

المناعة

التحليلات المرضية

الكيمياء الحيوية

مواضيع متنوعة أخرى

الانزيمات
Hydrophobic Effect
المؤلف:
P. R. Gibbs, A. Radzicka, and R. Wolfenden
المصدر:
J. Amer. Chem. Soc. 113, 4714–4715
الجزء والصفحة:
23-5-2016
3500
Hydrophobic Effect
Cooks and physicists are aware of the tendency of oil to separate from water, a solvent that is very self-cohesive. In biology, this effect is expressed in the tendency of uncharged (or nonpolar( molecules to escape from water by entering less polar surroundings, or by adhering to each other (Fig. 1). This phenomenon, termed the hydrophobic effect (1, 2), is believed to play a decisive role in maintaining the stability of biological membranes, in the proper folding of protein molecules, and in determining the relative affinities of hormones, antibodies, and substrates for proteins that bind them. The tendency of any particular nonpolar molecule or chemical group at equilibrium to favor transfer from water to a nonpolar phase, such as a hydrocarbon solvent, is termed its hydrophobicity.
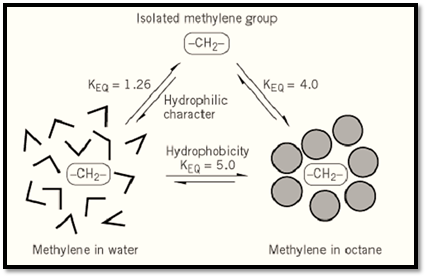
Figure 1. A partial view of the origins of the hydrophobic effect.
The physical origins of the hydrophobic effect remain controversial, because the properties of water as a solvent are not yet fully understood. It is of interest to ask whether nonpolar molecules, such as methane or ethane with no polar groups, tend to leave water and enter less polar solvents (octane is a familiar example) mainly because they are repelled by water; or whether they do so because they are attracted to the less polar solvent (or to each other). That question can be addressed by referring to an absolute standard, the vapor phase, that neither attracts nor repels solutes, and in which “solute” molecules exist in isolation most of the time. Using the vapor phase as a reference, single molecules of methane are found to exhibit an appreciable tendency to leave water, as indicated by their equilibria of transfer from water to the vapor phase. For methane, this equilibrium constant is 27 in favor of the vapor phase at 25°C. This tendency increases gradually with the size of a normal alkane, as can be seen in the series CH427, C 2H620, C3H829, C4H 1038, C5H1251, C6H1474, C7H1683. For each CH2 increment, transfer to the vapor phase is enhanced by an average factor of 1.26. In contrast, each CH 2 increment enhances the equilibrium of transfer from the vapor phase to a nonpolar solvent by a factor of 4.0. As a result, each CH 2 increment increases the distribution coefficient for transfer from water to a nonpolar solvent by a factor of 5.0 (Fig. 2). Similar behavior has been observed in the normal series of hydrocarbons, esters of acetic acid, amines, and alcohols (3) . In summary, single molecules of hydrocarbons have an appreciable tendency to leave water and enter the vapor phase, where they exist in isolation. These molecules are thus truly “hydrophobic” in the sense that they have an absolute tendency to escape from water. That is hardly surprising, since water is among the most self-cohesive molecules known, with an extremely high surface tension. However, this tendency to leave water is only slightly enhanced by increasing the size of the hydrocarbon (addition of a CH2 increment), whereas increasing size greatly enhances the tendency of a nonpolar solute to leave water and enter a nonpolar solvent. One is led to conclude that attraction to the nonpolar solvent also plays a major role in the hydrophobic effect, and this is believed to be due to van der Waals interactions, or London dispersion forces.
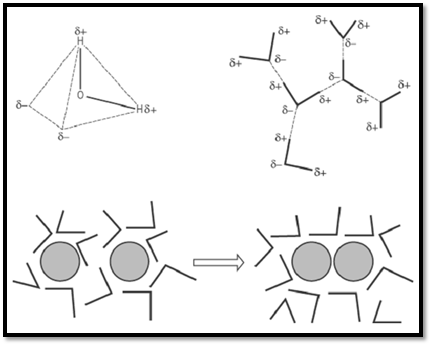
Figure 2. Analysis of the hydrophobic effect in terms of the equilibrium affinity of a solute for water and for a nonpolar liquid. ( a) Electrons are more strongly attracted by the oxygen atom of the water molecule, leaving partial negative charges near the corners of a tetrahedron, and partial positive charges at the other two corners. ( b) Water molecules are strongly attracted to each other by their partial charges, forming H-bonds between H and O atoms. As a result, liquid water is very self-cohesive. (c) Nonpolar molecules, like oil drops, break up the structure of water in which they are dissolved. The self-cohesiveness of water encourages these molecules or groups to coalesce, so that the part of the solvent can return to its preferred structure. This is called the hydrophilic effect.
The hydrophobic effect is unique, among the noncovalent forces involved in maintaining biological structures, in that it grows stronger with increasing temperature (1, 4), ie, enthalpy is released (DH is positive) when the force takes effect. If there is a net free energy of attraction (ie, DG is negative), then entropy must be released ( TDS must be positive and greater in magnitude than DH), since DG = DH–TDS. If a protein's stability were dependent entirely on the hydrophobic effect, then the protein would be expected to lose its structure at low temperatures, and some proteins do in fact undergo “cold denaturation”. There has been continuing interest in the probable structural origins of the entropy (and enthalpy) increases that accompany the formation of hydrophobic interactions. Changes in the properties of water in the immediate neighborhood of the solute almost certainly account, at least in part, for the loss of entropy that accompanies the introduction of a nonpolar molecule or methylene increment into water from the vapor phase or a nonpolar solvent. Frank and Evans (5) suggested that this might imply formation around solutes of a kind of clathrate or iceberg structure, in which water molecules were more ordered than in the bulk solute. These authors did not suggest that water surrounding a solute resembled Ice I in any literal sense, nor does current experimental evidence seem to support such a possibility. Entropy losses associated with introducing nonpolar solutes into water might also arise, at least in part, from restrictions on the mobility of solutes when they are introduced into a structured aqueous environment. In normal aliphatic compounds of increasing size, solubility might (according to this view) be reduced by progressive restrictions on internal rotation, and compounds with internal rotations already restricted would not be affected to the same extent. In apparent accord with this view, steroids and cycloalkanes display lower activity coefficients in water than nonrigid compounds of similar size, with reference to both nonpolar solvents and to the vapor phase (6), and the alicyclic amino acid proline is more hydrophilic than would be expected for an amino acid with an aliphatic side-chain of the same size (7). These effects could alternatively be due, at least in part, to the reduced surface area of cyclic as compared with open-chain compounds, resulting in the imposition of less severe restrictions on the solvent. These uncertainties illustrate the general difficulty of deconvoluting thermodynamic functions, which describe equations of state, without making assumptions that are themselves open to question. A clearer view of the origins of the structural origins of the hydrophobic effect awaits a more complete understanding of the properties of water and aqueous solutions.
All molecules, even those that are truly nonpolar, are attracted to each other by London dispersion forces. These forces are responsible for the fact that octane, and the lipid molecules present in biological membranes, are liquids at room temperature. They also make a major contribution to the hydrophobic effect discussed above, and to the resulting tendencies of protein molecules to adopt the specific structures needed for their activities. Anyone who has observed the wheels of a locomotive, or two cyclists riding a tandem bicycle, has a rough mental image of the London dispersion force. If
we picture two atoms of argon near each other, it is easy to imagine that temporary fluctuations in the distribution of electrons around one of these atoms may, at any given instant, place a partial negative charge toward the other atom. In response to this fluctuation, electrons in the orbitals of the neighboring atom tend to adjust slightly, placing a partial positive charge in the vicinity of the first atom and creating a small electrostatic attraction between them. A moment later, the roles may be reversed, but a net attraction remains. Because of the small charges involved, and their fluctuating character, London dispersion forces are individually weak and intensely dependent on the distances separating the atoms. These forces reach a maximum at an optimal distance equivalent to the sum of the van der Waals radii of the two atoms (typical van der Waals radii are 0.10 nm for H, 0.16 nm for C, 0.135 nm for O, 0.145 nm for N, and 0.17 nm for S). Beyond that distance, their mutual attraction falls off with the 6th power of the distance separating the two atoms. At distances closer than optimal, repulsion between the electron clouds of the two atoms builds up rapidly (unless they are able to share their electrons in a covalent bond), with the 12th power of the distance separating the atoms. The number and aggregate strength of the attractions between nonpolar molecules can be considerable, even though they are individually weak. They account for a substantial part of the hydrophobic effect discussed above and are thus largely responsible for maintaining the integrity of biological membranes and the proper folding of protein molecules.
References
1. W. Kauzmann (1959) Adv. Prot. Chem. 14, 1–45.
2. C. Tanford (1974) The Hydrophobic Effect, Wiley, New York, pp. 1–11.
3. R. Wolfenden and C. A. Lewis (1976) J. Theor. Biol. 59, 231–235.
4. G. Némethy and H. A. Scheraga (1962) J. Chem. Phys. 36, 3382–3400. 5. H. S. Frank and M. W. Evans (1945) J. Chem. Phys. 13, 507–532.
6. M. Osinga (1979) J. Amer. Chem. Soc. 101, 1621–1622.
7. P. R. Gibbs, A. Radzicka, and R. Wolfenden (1991) J. Amer. Chem. Soc. 113, 4714–4715.
 الاكثر قراءة في مواضيع عامة في الاحياء الجزيئي
الاكثر قراءة في مواضيع عامة في الاحياء الجزيئي
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















