

النبات

مواضيع عامة في علم النبات

الجذور - السيقان - الأوراق

النباتات الوعائية واللاوعائية

البذور (مغطاة البذور - عاريات البذور)

الطحالب

النباتات الطبية


الحيوان

مواضيع عامة في علم الحيوان

علم التشريح

التنوع الإحيائي

البايلوجيا الخلوية


الأحياء المجهرية

البكتيريا

الفطريات

الطفيليات

الفايروسات


علم الأمراض

الاورام

الامراض الوراثية

الامراض المناعية

الامراض المدارية

اضطرابات الدورة الدموية

مواضيع عامة في علم الامراض

الحشرات


التقانة الإحيائية

مواضيع عامة في التقانة الإحيائية


التقنية الحيوية المكروبية

التقنية الحيوية والميكروبات

الفعاليات الحيوية

وراثة الاحياء المجهرية

تصنيف الاحياء المجهرية

الاحياء المجهرية في الطبيعة

أيض الاجهاد

التقنية الحيوية والبيئة

التقنية الحيوية والطب

التقنية الحيوية والزراعة

التقنية الحيوية والصناعة

التقنية الحيوية والطاقة

البحار والطحالب الصغيرة

عزل البروتين

هندسة الجينات


التقنية الحياتية النانوية

مفاهيم التقنية الحيوية النانوية

التراكيب النانوية والمجاهر المستخدمة في رؤيتها

تصنيع وتخليق المواد النانوية

تطبيقات التقنية النانوية والحيوية النانوية

الرقائق والمتحسسات الحيوية

المصفوفات المجهرية وحاسوب الدنا

اللقاحات

البيئة والتلوث


علم الأجنة

اعضاء التكاثر وتشكل الاعراس

الاخصاب

التشطر

العصيبة وتشكل الجسيدات

تشكل اللواحق الجنينية

تكون المعيدة وظهور الطبقات الجنينية

مقدمة لعلم الاجنة


الأحياء الجزيئي

مواضيع عامة في الاحياء الجزيئي


علم وظائف الأعضاء


الغدد

مواضيع عامة في الغدد

الغدد الصم و هرموناتها

الجسم تحت السريري

الغدة النخامية

الغدة الكظرية

الغدة التناسلية

الغدة الدرقية والجار الدرقية

الغدة البنكرياسية

الغدة الصنوبرية

مواضيع عامة في علم وظائف الاعضاء

الخلية الحيوانية

الجهاز العصبي

أعضاء الحس

الجهاز العضلي

السوائل الجسمية

الجهاز الدوري والليمف

الجهاز التنفسي

الجهاز الهضمي

الجهاز البولي


المضادات الميكروبية

مواضيع عامة في المضادات الميكروبية

مضادات البكتيريا

مضادات الفطريات

مضادات الطفيليات

مضادات الفايروسات

علم الخلية

الوراثة

الأحياء العامة

المناعة

التحليلات المرضية

الكيمياء الحيوية

مواضيع متنوعة أخرى

الانزيمات
Purine Nucleotide Synthesis
المؤلف:
Denise R. Ferrier
المصدر:
Lippincott Illustrated Reviews: Biochemistry
الجزء والصفحة:
17-11-2021
3378
Purine Nucleotide Synthesis
The atoms of the purine ring are contributed by a number of compounds, including amino acids (aspartate, glycine, and glutamine), carbon dioxide (CO2), and N10-formyltetrahydrofolate (N10-formyl-THF), as shown in Figure 1. The purine ring is constructed primarily in the liver by a series of reactions that add the donated carbons and nitrogens to a preformed ribose 5-phosphate. [Note: Synthesis of ribose 5-phosphate from glucose 6-phosphate by the pentose phosphate pathway ]
Figure 1: Sources of the individual atoms in the purine ring. The order in which the atoms are added is shown by the numbers in the black boxes (see Fig. 3). CO2 = carbon dioxide.
A. 5-Phosphoribosyl-1-pyrophosphate synthesis
5-Phosphoribosyl-1-pyrophosphate (PRPP) is an activated pentose that participates in the synthesis and salvage of purines and pyrimidines. Synthesis of PRPP from ATP and ribose 5-phosphate is catalyzed by PRPP synthetase (Fig. 2). This X-linked enzyme is activated by inorganic phosphate and inhibited by purine nucleotides (end-product inhibition). [Note: Because the sugar moiety of PRPP is ribose, ribonucleotides are the end products of de novo purine synthesis. When deoxyribonucleotides are required for DNA synthesis, the ribose sugar moiety is reduced .]
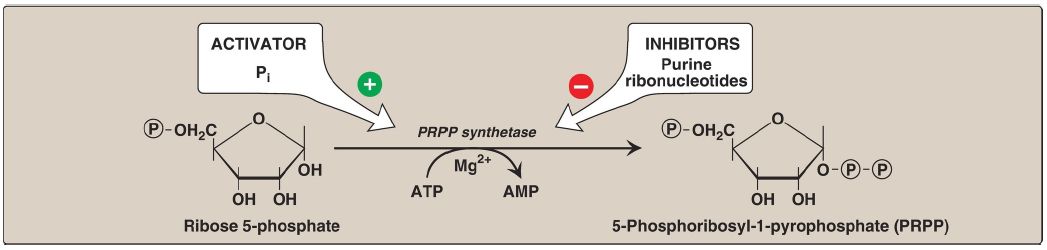
Figure 2: Synthesis of PRPP, showing the activator and inhibitors of the reaction. [Note: This is not the committed step of purine synthesis because PRPP is used in other pathways such as salvage (see p. 296).] = phosphate; Pi = inorganic phosphate; AMP = adenosine monophosphate; Mg = magnesium.
B. 5-Phosphoribosylamine synthesis
Synthesis of 5-phosphoribosylamine from PRPP and glutamine is shown in Figure 3. The amide group of glutamine replaces the pyrophosphate group attached to carbon 1 of PRPP. This is the committed step in purine nucleotide biosynthesis. The enzyme that catalyzes the reaction, glutamine:phosphoribosylpyrophosphate amidotransferase (GPAT), is inhibited by the purine 5′-nucleotides AMP and guanosine monophosphate (GMP, or guanylate), the end products of the pathway. The rate of the reaction is also controlled by the intracellular concentration of PRPP.
[Note: The concentration of PRPP is normally far below the Michaelis constant (Km) for the GPAT. Therefore, any small change in the PRPP concentration causes a proportional change in rate of the reaction .]

Figure 3: De novo synthesis of purine nucleotides, showing the inhibitory effect of some structural analogs. AMP and ADP = adenosine mono- and diphosphates; GMP = guanosine monophosphate; PRPP = 5-phosphoribosyl-1- pyrophosphate; Pi = inorganic phosphate; PPi = pyrophosphate; CO2 = carbon dioxide.
C. Inosine monophosphate synthesis
The next nine steps in purine nucleotide biosynthesis leading to the synthesis of inosine monophosphate ([IMP] whose base is hypoxanthine) are illustrated in Figure 22.7. IMP is the parent purine nucleotide for AMP and GMP. Four steps in this pathway require ATP as an energy source, and two steps in the pathway require N10-formyl-THF as a one-carbon donor . [Note: Hypoxanthine is found in tRNA .]
D. Synthetic inhibitors
Some synthetic inhibitors of purine synthesis (for example, the sulfonamides) are designed to inhibit the growth of rapidly dividing microorganisms without interfering with human cell functions (see Fig. 3). Other purine synthesis inhibitors, such as structural analogs of folic acid (for example, methotrexate), are used pharmacologically to control the spread of cancer by interfering with the synthesis of nucleotides and,
therefore, of DNA and RNA (see Fig. 3).
Inhibitors of human purine synthesis are extremely toxic to tissues, especially to developing structures such as in a fetus, or to cell types that normally replicate rapidly, including those of bone marrow, skin, gastrointestinal (GI) tract, immune system, or hair follicles. As a result, individuals taking such anticancer drugs can experience adverse effects, including anemia, scaly skin, GI tract disturbance, immunodeficiency, and hair loss.
E. Adenosine and guanosine monophosphate synthesis
The conversion of IMP to either AMP or GMP uses a two-step, energy- and nitrogen-requiring pathway (Fig. 4). [Note: AMP synthesis requires guanosine triphosphate (GTP) as an energy source and aspartate as a nitrogen source, whereas GMP synthesis requires ATP and glutamine.]
Also, the first reaction in each pathway is inhibited by the end product of that pathway. This provides a mechanism for diverting IMP to the synthesis of the purine present in lesser amounts. If both AMP and GMP are present in adequate amounts, the de novo pathway of purine nucleotide synthesis is inhibited at the GPAT step.
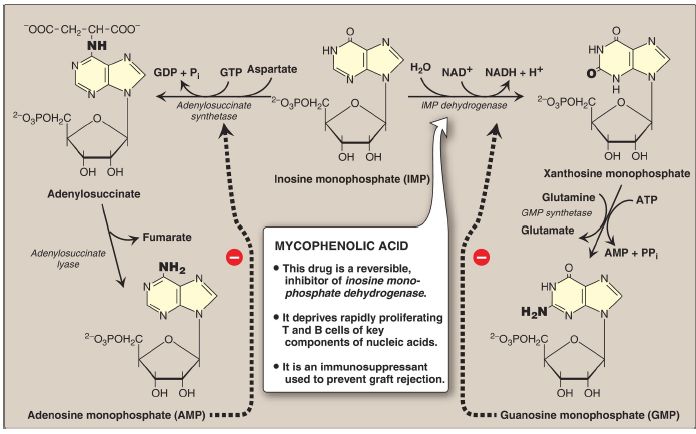
Figure 4: Conversion of IMP to AMP (or, adenylate) and GMP (or, guanylate) showing feedback inhibition. NAD(H) = nicotinamide adenine dinucleotide; GDP and GTP = guanosine di- and triphosphates; Pi = inorganic phosphate; PPi = pyrophosphate.
F. Nucleoside di- and triphosphate synthesis
Nucleoside diphosphates are synthesized from the corresponding nucleoside monophosphates by base-specific nucleoside monophosphate kinases (Fig. 5). [Note: These kinases do not discriminate between ribose or deoxyribose in the substrate.] ATP is generally the source of the transferred phosphate because it is present in higher concentrations than the other nucleoside triphosphates. Adenylate kinase is particularly active in the liver and in muscle, where the turnover of energy from ATP is high. Its function is to maintain equilibrium among the adenine nucleotides (AMP, ADP, and ATP). Nucleoside diphosphates and triphosphates are interconverted by nucleoside diphosphate kinase, an enzyme that, unlike the monophosphate kinases, has broad substrate specificity.
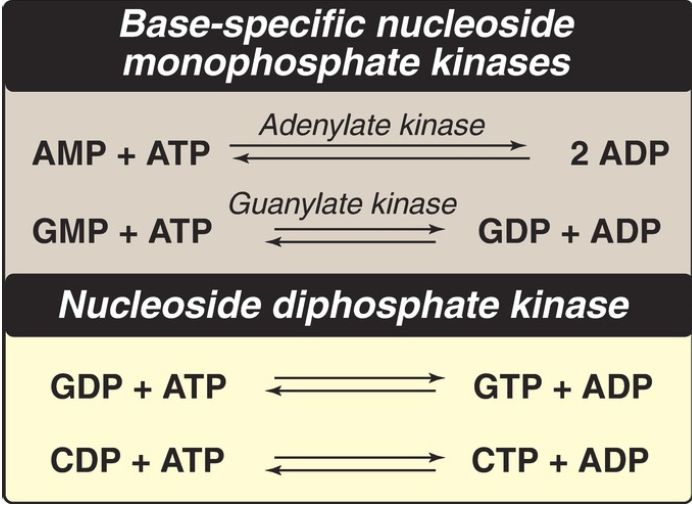
Figure 5: Conversion of nucleoside monophosphates to di- and triphosphates. AMP and ADP = adenosine mono- and diphosphates; GMP, GDP, and GTP = guanosine mono-, di-, and triphosphates; CDP and CTP = cytidine di- and triphosphates.
 الاكثر قراءة في الكيمياء الحيوية
الاكثر قراءة في الكيمياء الحيوية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















